For years, the conversation around amino acid dietary supplements was straightforward: build muscle, recover faster, repeat. Take your BCAAs, hit your protein targets, go again.
That model hasn’t just evolved — it’s been largely replaced.
In 2026, the questions being asked are different. Why are people on weight-loss medications losing strength even when the scale is dropping? Plant-based eaters can hit their daily protein targets and still fall short at a biological level. In current clinical and longevity research, muscle quality is becoming more important than muscle size.
The short answer is that muscle loss was never just about calories or protein totals. It’s about how your body processes, signals, and rebuilds tissue at the cellular level — and amino acids sit at the centre of all three.
What the Science Actually Says
Think of amino acid supplements as targeted metabolic inputs rather than protein shortcuts.
When you eat whole protein, digestion takes hours. Amino acids are released slowly into circulation. With free-form amino acids, absorption happens in roughly 15–30 minutes, blood levels spike rapidly, and signalling pathways — including muscle protein synthesis — activate faster. A 2013 study in the Journal of Nutrition confirmed that free-form EAAs stimulate muscle protein synthesis more efficiently than intact protein on an equivalent dose basis, particularly in older adults, where digestive efficiency is reduced.
That speed is why they’ve moved beyond gym culture into clinical nutrition, ageing protocols, and recovery under metabolic stress. They’re not replacing food — they’re correcting timing and composition gaps that food alone can’t always fill.
GLP-1 Drugs, Muscle Loss & Mitochondrial Remodelling
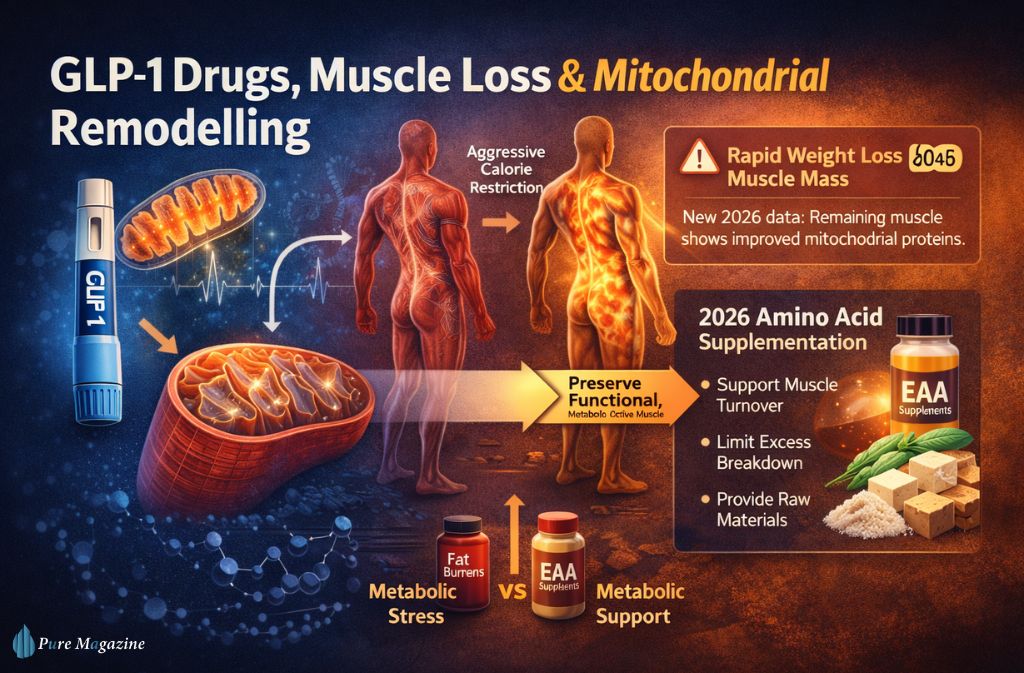
The early narrative around GLP-1 medications was simple: rapid weight loss means muscle loss, full stop. That framing is incomplete.
Emerging 2026 data tells a more nuanced story. Muscle mass may decrease during aggressive calorie restriction, but mitochondrial protein patterns inside the remaining muscle appear to improve. The muscle you keep becomes more metabolically efficient, even if there’s less of it by volume.
This matters for how amino acid supplementation is used in this context. The goal has shifted from building bigger muscles to preserving high-functioning, metabolically active tissue. EAAs support that by maintaining muscle protein turnover, limiting excessive breakdown during calorie restriction, and providing raw materials when appetite — and therefore food intake — is significantly suppressed.
If you’re also using other supplements alongside a GLP-1 protocol, it’s worth understanding how they interact. The conversation around stimulant-based fat burners, for instance, sits in a different category entirely from amino acid supplementation — one is driving metabolic stress, the other is helping the body manage it.
The Leucine Threshold — Updated
Leucine is the amino acid most directly responsible for triggering muscle protein synthesis. That much hasn’t changed. What’s shifted is the precision around dosing.
Current thinking puts the activation threshold at around 2.5–3g of leucine per meal. For people at elevated risk of sarcopenia — older adults, those in significant calorie deficits, people recovering from illness — roughly 6g daily is now suggested by emerging clinical research. A 2012 paper in the Journal of Physiology demonstrated that older muscle becomes increasingly resistant to normal protein signalling, requiring higher leucine doses to achieve the same anabolic response seen in younger adults.
The practical problem: you can eat enough total protein and still miss the leucine trigger consistently. A meal of 30g protein from mixed plant sources doesn’t necessarily deliver the leucine concentration needed to activate synthesis effectively. That’s where targeted EAA formulas outperform whole food alone — not because food is inferior, but because composition at the meal level is what actually matters.
Plant-Based Diets: The Meal-Level Gap
Most nutrition guidance focuses on daily protein totals. The more clinically relevant question in 2026 is meal-level amino acid adequacy — and this is where plant-based diets face a structural challenge that aggregate numbers hide.
A March 2026 analysis found that 56.6% of vegan meals were deficient in lysine. Not across the day — per meal. This isn’t a calorie problem or even a total protein problem. It’s a composition problem. Individual meals may lack the key amino acids needed to trigger repair, even when the day’s total looks fine on paper.
The practical fix isn’t necessarily a supplement. Structuring meals with a legume-to-grain ratio of roughly 2:1 goes a long way toward closing that gap naturally. But where that’s not consistently achievable, EAAs with lysine-heavy profiles function as a precision patch — targeted to what’s actually missing rather than blanketing everything.
This connects to a broader point about protein quality that comes up in conversations around women’s specific nutritional needs and protein for older adults — populations where amino acid adequacy, not just total intake, tends to be the limiting factor.
BCAAs: The U-Shaped Effect Nobody Talks About
BCAAs aren’t harmful. But they’re not context-free either.
Newer data have identified a U-shaped relationship between BCAA levels and metabolic outcomes, particularly in people with insulin resistance or Type 2 diabetes. Low BCAA levels are associated with poor muscle support. Moderate levels are beneficial. Excessive, sustained high doses may worsen insulin sensitivity — a finding consistent across several metabolic studies, including research published in Cell Metabolism linking chronically elevated circulating BCAAs to insulin resistance pathways.
For metabolically healthy people, BCAAs in normal doses are fine. For anyone managing blood sugar, full-spectrum EAAs are generally the smarter choice. The reasoning is straightforward: EAAs provide the complete signalling profile without the isolated BCAA load that appears problematic in certain metabolic contexts.
HMB: The Underrated Option
Not everyone wants to drink large volumes of amino acid supplements, particularly older adults or people in recovery. HMB — beta-hydroxy beta-methylbutyrate — is worth knowing about here.
It’s a metabolite of leucine, and clinically it’s shown consistent ability to reduce muscle protein breakdown. Roughly 3g of HMB produces an anti-catabolic effect comparable to much higher doses of leucine, making it a practical option when appetite or tolerance is limited. A meta-analysis in the Journal of the American Medical Directors Association found significant benefits in lean mass preservation among older adults supplementing with HMB — a population where muscle loss carries serious functional consequences.
Think of it as a low-volume muscle preservation tool rather than a performance supplement. The use cases are different, but so is the practicality.
Amino Acids and Longevity: Beyond the Muscle Conversation
The performance framing around amino acids is increasingly giving way to a longevity framing, and the underlying mechanisms are interesting.
Recent trials show that certain amino acid combinations can upregulate genes associated with energy production at the mitochondrial level and support DNA repair pathways. You won’t see immediate visible muscle gain from this. But cellular efficiency improves, and recovery capacity increases in ways that matter more over a ten-year horizon than over a ten-week training cycle.
This is why amino acids have started appearing in longevity protocols — not as muscle builders, but as metabolic support for ageing tissue. The same logic applies to nutrients like collagen, where the benefits are structural rather than performance-based. How collagen fits into longer-term tissue health is a separate conversation, but the underlying principle — supporting biological function at the cellular level rather than chasing visible short-term change — runs through both.
What to Look for on the Label
This is where quality separates from marketing.
Fermentation-derived amino acids are now the standard worth expecting. Lower-quality sources can be derived from animal byproducts, including feathers and hair — technically functional but ethically and qualitatively questionable. Vegan-fermented sources are cleaner and have become the default for better manufacturers.
Leucine content should be verifiable. Look for at least 2.5g per serving if muscle protein synthesis is your goal. If a product uses a proprietary blend that obscures individual amino acid amounts, that’s a reason to look elsewhere.
Third-party testing — NSF Certified for Sport or Informed-Sport — matters if you’re in any tested sport, and it’s a reasonable quality signal regardless. It means someone outside the manufacturer has verified what’s actually in the product.
Tracking your results over time is worth doing methodically. Understanding what blood markers can tell you about muscle metabolism and amino acid status gives you a way to assess whether supplementation is actually moving the needle, rather than just assuming it is.
Common Mistakes While Taking Amino Acid Supplements
Treating amino acids as a substitute for adequate protein intake is probably the most widespread one. They’re corrective tools, not replacements. Ignoring meal-level deficiencies while focusing on daily totals is the next — the aggregate number can look fine while individual meals are consistently missing key triggers. Overusing isolated BCAAs without accounting for metabolic context is increasingly recognised as a real risk rather than a theoretical one. And buying products with proprietary blends that hide individual amino acid doses makes it impossible to know whether you’re actually hitting the thresholds that matter.
Quick Decision Framework
Amino acid supplementation makes practical sense if you’re on a GLP-1 medication, over 40 and noticing strength or muscle changes, following a plant-based diet with variable protein quality, or in a sustained calorie deficit for weight loss purposes.
Be more strategic — and lean toward EAAs over BCAAs — if you’re managing blood sugar or insulin sensitivity.
Skip them if your protein intake is consistent, complete, and varied. There’s no meaningful benefit to supplementing adequacy.
FAQs
Q. Can I take amino acids on GLP-1 medications?
Yes — amino acid supplements, especially essential amino acids (EAAs), can be helpful while using GLP-1 medications. They support muscle preservation and protein turnover when appetite is reduced and protein intake drops. In 2026, they’re commonly used to protect lean muscle during rapid weight loss.
Q. What is the leucine trigger?
The leucine trigger is the minimum amount of leucine needed to activate muscle protein synthesis. Research shows this is around 2.5–3 grams per meal. If you don’t reach this level, your body may not fully use the protein you consume for muscle repair.
Q. How much leucine do I need daily?
For individuals at higher risk of muscle loss (such as older adults or those in a calorie deficit), emerging research suggests around 6 grams of leucine per day. For healthy individuals with adequate protein intake, daily leucine intake is usually covered through diet.
Q. Are BCAA supplements safe for people with diabetes?
BCAAs are generally safe in moderate amounts. However, consistently high doses may negatively affect insulin sensitivity. For people managing blood sugar levels, full-spectrum essential amino acids (EAAs) are typically a safer and more balanced option.
Q. Do plant-based diets require amino acid supplements?
Not always, but plant-based diets often have meal-level amino acid gaps, especially in lysine. Amino acid supplements can help fill these gaps more efficiently than relying only on food combinations, particularly in low-calorie or restrictive diets.
Q. What is HMB, and does it work for muscle preservation?
HMB (β-Hydroxy β-Methylbutyrate) is a compound derived from leucine that helps reduce muscle breakdown. It is especially useful for older adults, people recovering from illness, or anyone unable to consume enough protein. Clinical evidence supports its role in preserving lean muscle mass.
Q. Do amino acid supplements help with muscle recovery?
Yes — amino acid dietary supplements can improve recovery after exercise by supporting muscle repair and reducing breakdown. Essential amino acids are more effective than BCAAs alone because they provide the full profile needed for protein synthesis.
Q. When is the best time to take amino acid supplements?
The most effective times are:
- Before or during workouts (to reduce muscle breakdown)
- After exercise (to support recovery)
- Between meals (if protein intake is low)
Timing matters less than meeting amino acid needs consistently.
The Bottom Line
Amino acid dietary supplements have moved well past the gym-culture framing they came from. In 2026, the relevant conversations are about muscle quality over size, mitochondrial health, meal-level composition gaps, and long-term metabolic resilience.
Used with that understanding, they’re genuinely useful tools for specific situations. Used as a blanket daily habit without context, they’re just expensive redundancy.
The edge comes from knowing which situation you’re actually in.
Related: Supplement Jodium: Benefits, Dosage & Hidden Risks (2026)
|
Disclaimer: This article is for informational purposes only and does not constitute medical advice. Always consult a qualified healthcare professional before starting any new supplement regimen. |